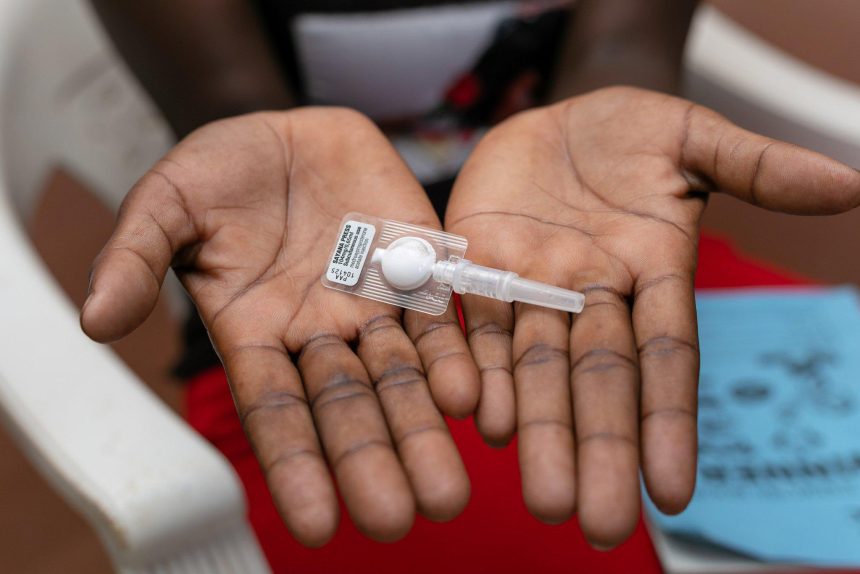
The Ministry of Health, in partnership with Phillips Pharmaceuticals (Uganda) Ltd,(PPUL) PATH Advocacy, and Population Services International (PSI) Uganda, has launched the SAYANA PRESS single-unit injectable contraceptive for use in the private sector in a major step toward improving reproductive health access.
The initiative aligns with Uganda’s National Guidelines on Self-Care Interventions for Health, which stress the importance of accessible and affordable self-care options in empowering individuals to make informed decisions about their health.

SAYANA PRESS is a long-term contraceptive designed for self-administration following consultation with a qualified healthcare provider. It offers a convenient and discreet option for women seeking to manage their reproductive health independently, especially in settings where access to healthcare facilities may be limited.
Dr. Charles Olaro, Director General of Health Services, emphasized the importance of community involvement: “As we roll out SAYANA PRESS, we must explore ways to effectively engage men—especially husbands—as supportive partners in family planning.”

The availability of SAYANA PRESS in the private sector marks a significant milestone in Uganda’s journey toward inclusive, self-directed health solutions.
“This product represents more than just a new contraceptive method—it’s a tool of empowerment,” said Dr. Richard Mugahi, Commissioner for Reproductive, Maternal and Child Health.
“With 45% of pregnancies in Uganda being unintended, we are working to diversify family planning options and ensure they are within reach when needed,”he added.
The launch highlights a strong public-private collaboration: Phillips Pharmaceuticals will handle product distribution, PATH is supporting the training of healthcare providers, and PSI Uganda is leading efforts in market development and promoting women’s empowerment through education and access.
Ani Prajith,CEO,PPUL noted that the initiative is expected to contribute to a reduction in unintended pregnancies, while advancing the country’s broader goals for universal access to sexual and reproductive health services.

“The whole approach is to see how we can bring into a market access program wherein it can be brought into an affordable model because now the people are able to get the product from the private pharmacies and the clinics across the country. Philips is known to be one of the largest distributors in terms of having a last mile distribution network where we are able to deliver this product across the 45 million population through several healthcare outlets,”he noted.
For the past couple of years PSI has been leading the implementation of a DMPAI-SC project which is delivering innovations in self-care and this specifically focused on promoting family planning in the context of choice for women but specifically focusing on a method which enables women to self-inject every three months.

Stephen Alege,Lead for the new DMPA-SC private sector says ,”this is very critical because it puts power in the hands of the women, they are able to utilize family planning wherever they are, when they want, how they want and we’ve been a part of this largely in the public sector.”
Manufactured by Pfizer and scientifically known as Medroxyprogesterone contraceptive injections , its other brand name is Depo-Provera.
Like all medicines, medroxyprogesterone contraceptive injections can cause side effects although not everyone gets them, according to the National Health Service-UK since they happen in more than 1 in 100 people.